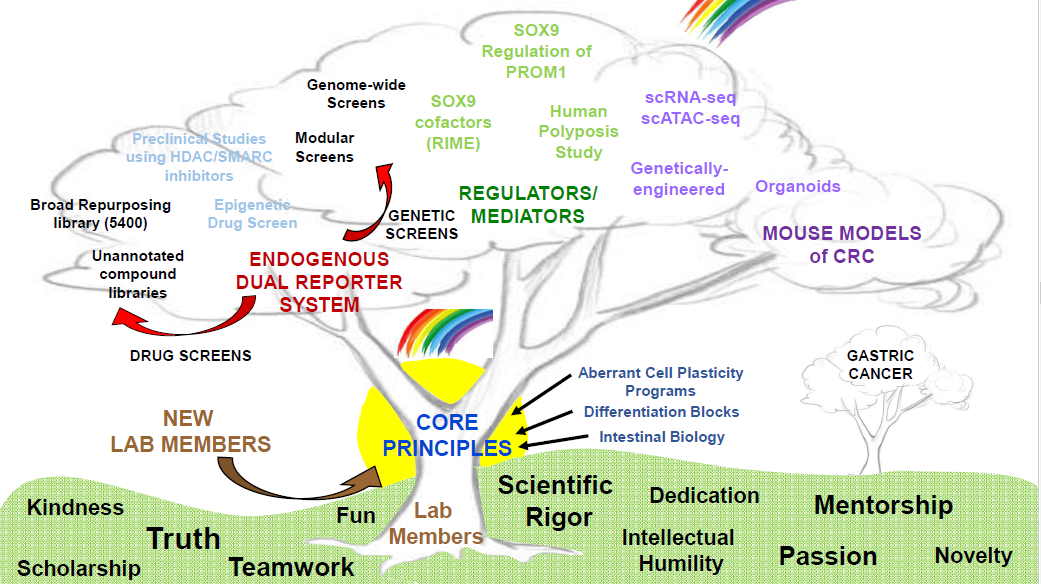
RESEARCH PROGRAM
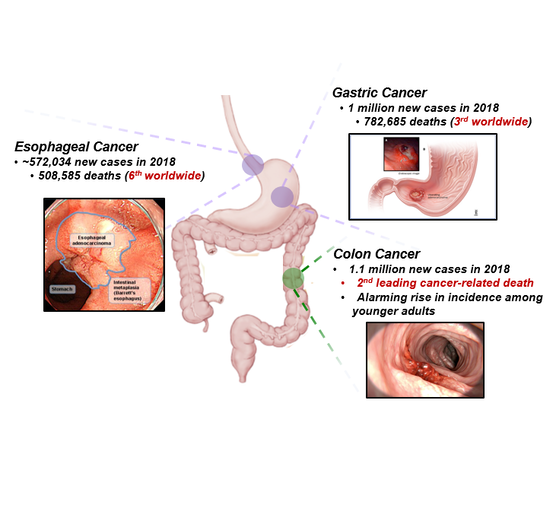
Gastric and esophageal cancers: We are devoted to better understanding cancers of the stomach and lower esophagus. Using model systems that integrate disease risk factors with early genetic mistakes, we aim to study the premalignant state, hoping to learn how somatic mutations engage environmental exposures during the initiation of these cancers. These models have yielded new insight into disease progression, revealing therapeutic vulnerabilities that are actively being investigated in preclinical systems. By leveraging data from patients, mouse models, and cell culture studies, we are motivated to provide an integrated understanding of early events in gastrointestinal cancers with the ultimate hope that the knowledge gained will inspire new avenues for cancer prevention and treatment.
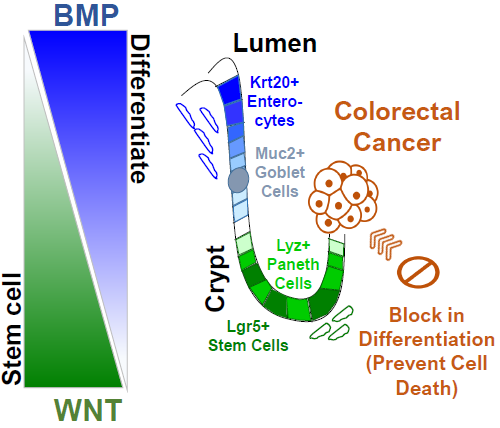
Colorectal cancer: Our lab is invested in a deeper understanding of colorectal cancer, which remains the second leading cause of cancer-mortality worldwide and responsible for an alarming trend of increasing incidence in younger patients. The intestine is a special organ in that it is the most rapidly renewing tissue in our body; the gut lining is replenished every 5 to 7 days, which is simply remarkable. Maintaining the intestinal lining therefore requires stem cells to constantly give rise to mature cells to perform their specialized function before dying. Disruption of this natural maturation is a hallmark of colorectal cancer. Our lab is focused on defining the molecular components that block proper differentiation in cancer cells. We have also designed a unique discovery platform to help us screen chemical compound libraries with the goal of identifying specific agents that overcome this block in maturation. We strongly believe the next generation of therapeutics will restore proper maturation in colon cancer cells, reprogramming them to live out their natural life cycle. We have also noticed that factors responsible for stem cell behavior are mutated in a subset of colorectal cancer. Using innovative mouse models and cutting-edge technology, we hope to define the clinical and functional significance of these recurrent mutations.
Sethi Lab - Gastrointestinal Malignancies
HOME | RESEARCH | PEOPLE | PUBLICATIONS | SUPPORT | CONTACT
©2023 Sethi Lab at Dana-Farber Cancer Institute
©2023 Sethi Lab at Dana-Farber Cancer Institute